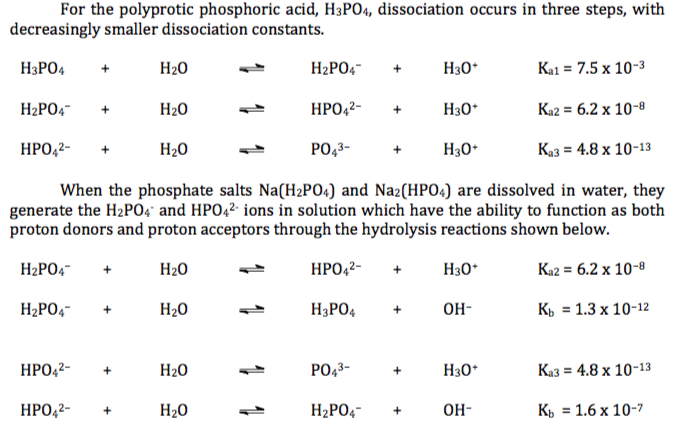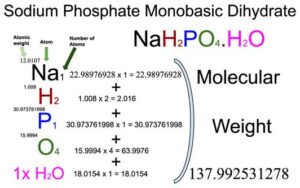
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes

El monohidrato de fosfato de sodio Dihyrogen Nah2PO4· H2O. - China Mono fosfato de sodio, Metal detergente
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%201KG.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs
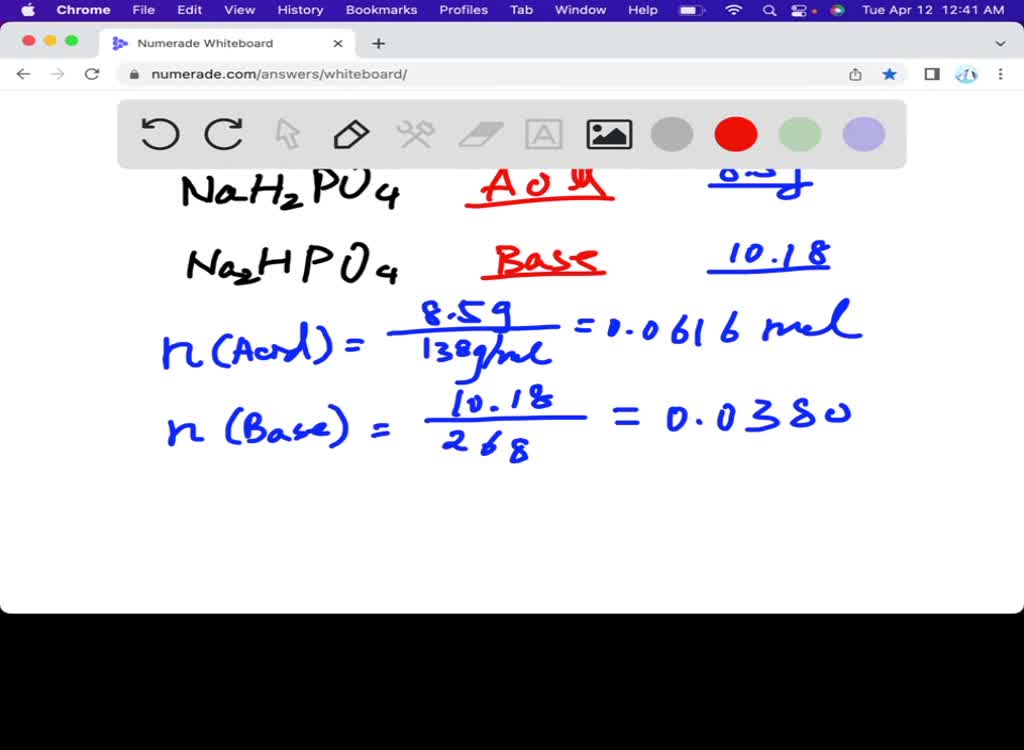
SOLVED: Calculate the mass of NaH2PO4 ∙ H2O (in grams) required to prepare 250mL of pH 7.21 buffer solution if the total buffer concentration is to be 0.20M. The Ka of NaH2PO4∙H2O

Jual Sodium dihydrogen phosphate monohydrate/Nah2PO4.h2O - Kab. Bantul - muda berkah jogja | Tokopedia
![Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific](https://m.media-amazon.com/images/I/71S3q3Er55L.jpg)
Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen Phosphate monohydrate], 1 Kilogram: Amazon.com: Industrial & Scientific

10049-21-5, 137.99, Sodium Phosphate, Monobasic, Monohydrate, Crystal, Reagent, ACS - 6NNX9|S1395-500GM - Grainger
Welcome to Chem Zipper.com......: In a determination of P an aqueous solution of NaH2PO4 is treated with a mixture of Ammonium and magnesium ions to precipitate magnesium Ammonium phosphate (Mg(NH4)PO4.6H2O. This is
![S23120-5000.0 - Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen phosphate monohydrate], 5 Kilograms S23120-5000.0 - Sodium Phosphate Monobasic Monohydrate [Sodium dihydrogen phosphate monohydrate], 5 Kilograms](https://d2gdaxkudte5p.cloudfront.net/system/images/plabel_14792_20220712-103504.jpg)